Yersinia pestis is the causative agent of plague. What do we know about the causative agent of the plague?
More than half of the population of Europe in the Middle Ages (XIV century) was wiped out by a plague known as the black death. The horror of these epidemics remained in the memory of people after several centuries and was even captured in the canvases of artists. Further, the plague repeatedly visited Europe and claimed human lives, albeit not in such quantities.
Currently, the plague remains a disease. About 2 thousand people become infected annually. Of them most of dies. Most cases of infection are observed in the northern regions of China and the countries of Central Asia. According to experts, there are no reasons and conditions for the appearance of black death today.
The causative agent of the plague was discovered in 1894. Studying the epidemics of the disease, Russian scientists developed the principles of the development of the disease, its diagnosis and treatment, and an anti-plague vaccine was created.
Symptoms of plague depend on the form of the disease. When the lungs are affected, patients become highly contagious, as the infection spreads into the environment by airborne droplets. With the bubonic form of plague, patients are slightly contagious or not contagious at all. In the secretions of the affected lymph nodes, pathogens are absent, or there are very few of them.
Plague treatment has become much more effective with the advent of modern antibacterial drugs. Mortality from the plague has since dropped to 70%.
Plague prevention includes a number of measures that limit the spread of infection.
Plague is an acute infectious zoonotic vector-borne disease, which in the CIS countries, together with diseases such as cholera, tularemia and smallpox, is considered (OOI).
Rice. 1. Painting “The Triumph of Death”. Pieter Bruegel.
Plague agent
In 1878, G. N. Minkh and in 1894, A. Yersin and S. Kitazato, independently discovered the causative agent of the plague. Subsequently, Russian scientists studied the mechanism of development of the disease, the principles of diagnosis and treatment, and created an anti-plague vaccine.
- The causative agent of the disease (Yersinia pestis) is a bipolar, non-motile coccobacillus, which has a delicate capsule and never forms spores. The ability to form a capsule and antiphagocytic mucus does not allow macrophages and leukocytes to actively fight the pathogen, as a result of which it quickly multiplies in the organs and tissues of humans and animals, spreading through the bloodstream and through the lymphatic tract throughout the body.
- Plague pathogens produce exotoxins and endotoxins. Exo- and endotoxins are contained in the bodies and capsules of bacteria.
- Enzymes of bacterial aggression (hyaluronidase, coagulase, fibrinolysin, hemolysin) facilitate their penetration into the body. The stick is able to penetrate even intact skin.
- In the ground, the plague bacillus does not lose its viability for up to several months. It survives in the corpses of animals and rodents for up to one month.
- Bacteria are resistant to low temperatures and freezing.
- Plague pathogens are sensitive to high temperatures, acidic environment and sun rays, which kill them in only 2 - 3 hours.
- Pathogens are stored in pus for up to 30 days, in milk for up to 3 months, and in water for up to 50 days.
- Disinfectants destroy the plague bacillus in a few minutes.
- Plague pathogens cause disease in 250 animal species. Among them, the majority are rodents. Camels, foxes, cats and other animals are susceptible to the disease.

Rice. 2. In the photo, the plague bacillus is the bacterium that causes the plague - Yersinia pestis.
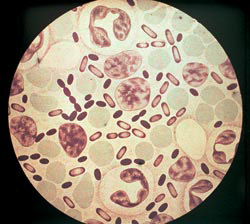
Rice. 3. The photo shows the causative agents of the plague. The intensity of coloration with aniline dyes is greatest at the poles of bacteria.
Php?post=4145&action=edit#
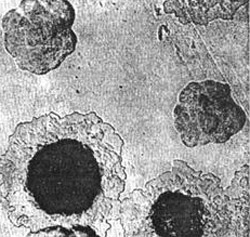
Rice. 4. In the photo, the pathogens of the plague are growing on a dense colony medium. At first, the colonies look like broken glass. Next, their central part becomes denser, and the periphery resembles lace.
Epidemiology
Reservoir of infection
Rodents (tarbagans, marmots, gerbils, gophers, rats and house mice) and animals (camels, cats, foxes, hares, hedgehogs, etc.) are easily susceptible to the plague bacillus. Among laboratory animals, white mice are susceptible to infection, Guinea pigs, rabbits and monkeys.
Dogs never get plague, but they transmit the pathogen through the bites of blood-sucking insects - fleas. An animal that dies from a disease ceases to be a source of infection. If rodents infected with plague bacilli hibernate, their disease becomes latent, and after hibernation they again become distributors of pathogens. In total, there are up to 250 species of animals that are sick, and therefore are a source and reservoir of infection.

Rice. 5. Rodents are the reservoir and source of the plague pathogen.

Rice. 6. The photo shows signs of plague in rodents: enlarged lymph nodes and multiple hemorrhages under the skin.

Rice. 7. In the photo, the small jerboa is a carrier of the plague in Central Asia.

Rice. 8. In the photo, the black rat is a carrier not only of plague, but also of leptospirosis, leishmaniasis, salmonellosis, trichinosis, etc.
Routes of infection
- The main route of transmission of pathogens is through flea bites (transmissible route).
- The infection can enter the human body when working with sick animals: slaughter, skinning and cutting (contact route).
- Pathogens can enter the human body with contaminated food products as a result of their insufficient heat treatment.
- From a patient with pulmonary form Plague infection is spread by airborne droplets.

Rice. 9. The photo shows a flea on human skin.

Rice. 10. The photo shows the moment of a flea bite.
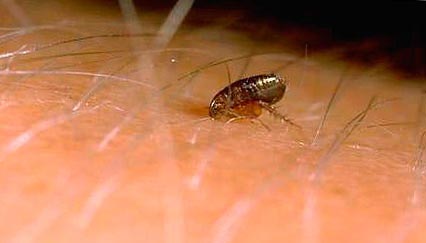
Rice. 11. The moment of a flea bite.
Pathogen carriers
- The carriers of pathogens are fleas (in nature there are more than 100 species of these arthropod insects),
- Some types of ticks are carriers of pathogens.
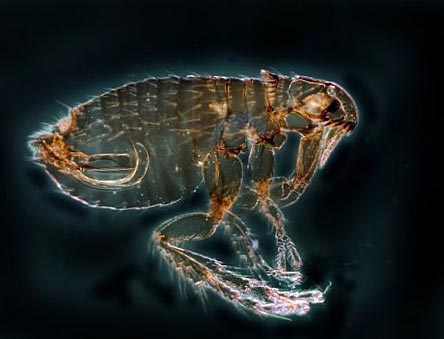
Rice. 12. In the photo, the flea is the main carrier of the plague. There are more than 100 species of these insects in nature.

Rice. 13. In the photo, the gopher flea is the main carrier of plague.
How does infection occur?
Infection occurs through an insect bite and rubbing of its feces and intestinal contents when regurgitating during feeding. When bacteria multiply in the intestinal tube of a flea under the influence of coagulase (an enzyme secreted by pathogens), a “plug” is formed that prevents human blood from entering its body. As a result, the flea regurgitates a clot onto the skin of the bitten person. Infected fleas remain highly infective for 7 weeks to 1 year.

Rice. 14. In the photo, the appearance of a flea bite is pulicotic irritation.
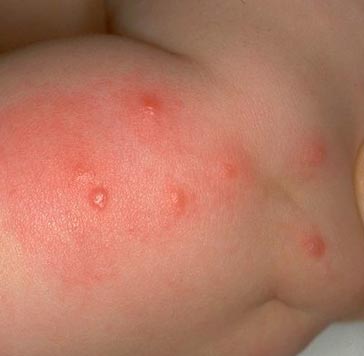
Rice. 15. The photo shows a characteristic series of flea bites.

Rice. 16. View of the lower leg with flea bites.

Rice. 17. Appearance of the thigh with flea bites.
Man as a source of infection
- When the lungs are affected, patients become highly contagious. The infection spreads into the environment through airborne droplets.
- With the bubonic form of plague, patients are slightly contagious or not contagious at all. There are no pathogens in the secretions of the affected lymph nodes, or there are very few of them.
Mechanisms of plague development
The ability of the plague bacillus to form a capsule and antiphagocytic mucus does not allow macrophages and leukocytes to actively fight it, as a result of which the pathogen quickly multiplies in the organs and tissues of humans and animals.
- Plague pathogens penetrate through damaged skin and then through the lymphatic tract into the lymph nodes, which become inflamed and form conglomerates (buboes). Inflammation develops at the site of the insect bite.
- Penetration of the pathogen into the bloodstream and its massive reproduction leads to the development of bacterial sepsis.
- From a patient with pneumonic plague, the infection spreads through airborne droplets. The bacteria enter the alveoli and cause severe pneumonia.
- In response to the massive multiplication of bacteria, the patient's body produces a huge number of inflammatory mediators. Developing disseminated intravascular coagulation syndrome(DIC syndrome), in which everyone is affected internal organs. Of particular danger to the body are hemorrhages in the heart muscle and adrenal glands. The developed infectious-toxic shock causes the death of the patient.
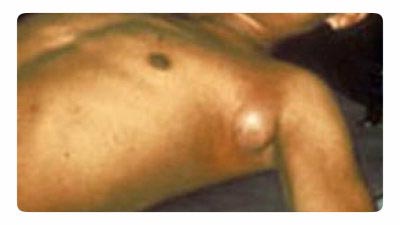
Rice. 18. The photo shows the bubonic plague. Typical enlargement of the lymph node in the axillary area.
Plague symptoms
The disease manifests itself after the penetration of the pathogen into the body for 3-6 days (rarely, but there have been cases of manifestation of the disease on the 9th day). When an infection enters the bloodstream, the incubation period is several hours.
Clinical picture of the initial period
- Acute onset, high temperatures and chills.
- Myalgia (muscle pain).
- Excruciating thirst.
- A strong sign of weakness.
- Rapid development of psychomotor agitation (“such patients are called crazy”). A mask of horror (“plague mask”) appears on the face. Lethargy and apathy are less common.
- The face becomes hyperemic and puffy.
- The tongue is thickly covered with a white coating (“chalky tongue”).
- Multiple hemorrhages appear on the skin.
- The heart rate increases significantly. Arrhythmia appears. Blood pressure drops.
- Breathing becomes shallow and rapid (tachypnea).
- The amount of urine excreted decreases sharply. Anuria develops (complete absence of urine output).
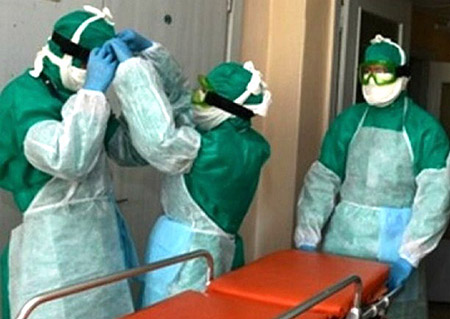
Rice. 19. In the photo, assistance to a plague patient is provided by doctors dressed in anti-plague suits.
Forms of plague
Local forms of the disease
Cutaneous form
At the site of a flea bite or contact with an infected animal on skin a papule appears, which quickly ulcerates. Next, a black scab and scar appear. Most often, skin manifestations are the first signs of more severe manifestations of the plague.
Bubonic form
The most common form of manifestation of the disease. An increase in lymph nodes appears near the site of an insect bite (inguinal, axillary, cervical). More often one lymph node becomes inflamed, less often several. When several lymph nodes become inflamed at once, a painful bubo is formed. Initially, the lymph node has a hard consistency, painful on palpation. Gradually it softens, acquiring a dough-like consistency. Next, the lymph node either resolves or becomes ulcerated and sclerosed. From the affected lymph node, the infection can enter the bloodstream, with the subsequent development of bacterial sepsis. Acute phase The bubonic form of plague lasts about a week.

Rice. 20. The photo shows the affected cervical lymph nodes (buboes). Multiple hemorrhages of the skin.
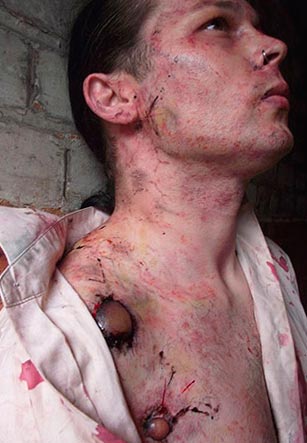
Rice. 21. In the photo, the bubonic form of plague is a defeat cervical lymph nodes. Multiple hemorrhages in the skin.

Rice. 22. The photo shows the bubonic form of plague.
Common (generalized) forms
When the pathogen enters the bloodstream, widespread (generalized) forms of plague develop.
Primary septic form
If the infection, bypassing the lymph nodes, immediately enters the blood, then the primary septic form of the disease develops. Intoxication develops at lightning speed. With the massive proliferation of pathogens in the patient’s body, a huge number of inflammatory mediators are produced. This leads to the development of disseminated intravascular coagulation syndrome (DIC), which affects all internal organs. Hemorrhages in the heart muscle and adrenal glands pose a particular danger to the body. The developed infectious-toxic shock causes the death of the patient.
Secondary septic form of the disease
When the infection spreads beyond the affected lymph nodes and pathogens enter the bloodstream, infectious sepsis develops, which manifests itself sharp deterioration the patient's condition, increased symptoms of intoxication and the development of DIC syndrome. The developed infectious-toxic shock causes the death of the patient.

Rice. 23. In the photo, the septic form of plague is the consequences of disseminated intravascular coagulation syndrome.

Rice. 24. In the photo, the septic form of plague is the consequences of disseminated intravascular coagulation syndrome.
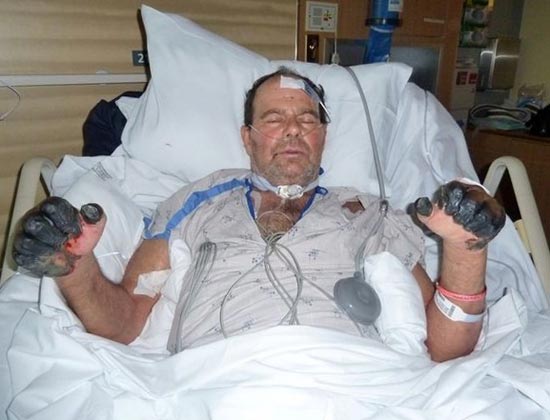
Rice. 25. 59-year-old Paul Gaylord (resident of Portland, Oregon, USA). Plague bacteria entered his body from a stray cat. As a result of the development of a secondary septic form of the disease, his fingers and toes were amputated.

Rice. 26. Consequences of DIC syndrome.
Externally disseminated forms of the disease
Primary pulmonary form
The pneumonic form of plague is the most severe and dangerous form of the disease. The infection penetrates the alveoli through airborne droplets. Damage to the lung tissue is accompanied by cough and shortness of breath. An increase in body temperature occurs with severe chills. The sputum at the beginning of the disease is thick and transparent (vitreous), then it becomes liquid and foamy, mixed with blood. The scant data from physical examinations do not correspond to the severity of the disease. DIC syndrome develops. Internal organs are affected. Hemorrhages in the heart muscle and adrenal glands pose a particular danger to the body. The death of the patient occurs from infectious-toxic shock.
When the lungs are affected, patients become highly contagious. They form around themselves a focus of a particularly dangerous infectious disease.
Secondary pulmonary form
It is an extremely dangerous and severe form of the disease. Pathogens penetrate the lung tissue from affected lymph nodes or through the bloodstream during bacterial sepsis. The clinic and the outcome of the disease, as in the primary pulmonary form.
Intestinal form
The existence of this form of the disease is controversial. It is assumed that infection occurs through the consumption of contaminated products. Initially, against the background of intoxication syndrome, abdominal pain and vomiting appear. Then diarrhea and numerous urges (tenesmus) join. The stool is profuse, mucosal-bloody.
Rice. 27. Photo of an anti-plague suit - special equipment for medical workers when eliminating an outbreak of a particularly dangerous infectious disease.
Laboratory diagnosis of plague
The basis for the diagnosis of plague is the rapid detection of the plague bacillus. First, bacterioscopy of smears is performed. Next, a culture of the pathogen is isolated, which infects experimental animals.
The material for research is the contents of the bubo, sputum, blood, feces, pieces of tissue from the organs of deceased animals and corpses.
Bacterioscopy
The causative agent of plague (Yersinia pestis) is a rod-shaped bipolar coccobacilli. Analysis for the detection of plague bacillus by direct bacterioscopy is the simplest and in a fast way. The waiting time for the result is no more than 2 hours.
Crops of biological material
The culture of the plague pathogen is isolated in specialized high-security laboratories designed to work with. The growth time of the pathogen culture is two days. Next, an antibiotic sensitivity test is performed.
Serological methods
The use of serological methods makes it possible to determine the presence and growth of antibodies in the patient’s blood serum to the plague pathogen. The time to receive the result is 7 days.

Rice. 28. Diagnosis of plague is carried out in special sensitive laboratories.

Rice. 29. The photo shows the causative agents of the plague. Fluorescence microscopy.

Rice. 30. The photo shows the culture of Yersinia pestis.
Immunity to plague
Antibodies against the introduction of the plague pathogen are formed in sufficient time late dates development of the disease. Immunity after an illness is not long-lasting or intense. There are repeated cases of the disease, which are as severe as the first.
Treatment of plague
Before treatment begins, the patient is hospitalized in a separate room. Medical staff, serving the patient, dresses in a special anti-plague suit.
Antibacterial treatment
Antibacterial treatment begins at the first signs and manifestations of the disease. Among antibiotics, preference is given to antibacterial drugs aminoglycoside groups (streptomycin), tetracycline groups (vibromycin, morphocycline), fluoroquinolone groups (ciprofloxacin), ansamycin groups (rifampicin). The antibiotic of the amphenicol group (cortrimoxazole) has proven itself in the treatment of the skin form of the disease. For septic forms of the disease, a combination of antibiotics is recommended. The course of antibacterial therapy is at least 7–10 days.
Treatment aimed at different stages of development of the pathological process
The goal of pathogenetic therapy is to reduce the intoxication syndrome by removing toxins from the patient's blood.
- The introduction of fresh frozen plasma, protein preparations, rheopolyglucin and other drugs in combination with forced diuresis is shown.
- Improvement of microcirculation is achieved by using trental in combination with salcoseryl or picamilon.
- With the development of hemorrhages, plasmapheresis is immediately performed in order to stop the syndrome of disseminated intravascular coagulation.
- If blood pressure drops, dopamide is prescribed. This condition indicates the generalization and development of sepsis.
Symptomatic treatment
Symptomatic treatment is aimed at suppressing and eliminating the manifestations (symptoms) of the plague and, as a result, alleviating the suffering of the patient. It is aimed at eliminating pain, cough, shortness of breath, suffocation, tachycardia, etc.
The patient is considered healthy if all symptoms of the disease have disappeared and 3 negative results bacteriological research.
Anti-epidemic measures
Identification of a plague patient is a signal for immediate action, which includes:
- carrying out quarantine measures;
- immediate isolation of the patient and carrying out preventive antibacterial treatment service personnel;
- disinfection at the source of the disease;
- vaccination of persons in contact with the patient.
After vaccination with an anti-plague vaccine, immunity lasts for a year. Re-vaccinate after 6 months. persons at risk reinfection: shepherds, hunters, agricultural workers and employees of anti-plague institutions.

Rice. 31. In the photo, the medical team is dressed in anti-plague suits.
Disease prognosis
The prognosis of plague depends on the following factors:
- forms of the disease,
- timeliness of treatment started,
- availability of the entire arsenal of medicinal and non-medicinal treatments.
The most favorable prognosis in patients with lesions of the lymph nodes. Mortality in this form of the disease reaches 5%. In the septic form of the disease, the mortality rate reaches 95%.
The plague is, and even with the application of all necessary medicines and manipulation, the disease often ends in the death of the patient. Plague pathogens constantly circulate in nature and cannot be completely destroyed and controlled. Symptoms of plague are varied and depend on the form of the disease. The bubonic form of plague is the most common.
Articles in the section "Particularly dangerous infections"Most popular
Article for the “bio/mol/text” competition: At first glance, it is a microscopic and harmless bacterium, but in reality it is a merciless killer that killed almost a third of the European population in the 14th century. Yersinia pestis, popularly known as plague stick, is the causative agent of the most dangerous disease - plague. Since ancient times around Y. pestis Scientific disputes arose that continue to this day. The most striking of them, as well as facts from the life of one of the most mysterious microorganisms, are highlighted in this article.
Note!
This work took first place in the “Best News Message” category of the “bio/mol/text” 2015 competition.
The sponsor of the nomination “Best article on the mechanisms of aging and longevity” is the Science for Life Extension Foundation. The audience award was sponsored by Helicon.
Competition sponsors: Biotechnology Research Laboratory 3D Bioprinting Solutions and Scientific Graphics, Animation and Modeling Studio Visual Science.
Stranger in a mask
Y. pestis was hidden from human eyes for a long time due to many circumstances. At first, size was an obstacle: before the invention of the microscope by the Jansen brothers in 1590* and its further use by Hooke and Leeuwenhoek (to whom, by the way, bacteria and protozoa were born), no one even thought about the fact that in addition to living objects visible to the eye There are also tiny organisms. But even with the advent of optics, the plague wand continued to maintain its incognito status, blaming the bacteria of the birth Bacterium, Bacillus And Pasteurella. Meanwhile Y. pestis freely walked around the world and carried away human lives more than war.
There have been three plague pandemics in history. The first of these was the “Justinian Plague” (named after the Byzantine ruler Justinian I), which swept through the entire civilized world of that time. Historians date it back to 541-580 AD, although outbreaks of an unknown disease plagued humanity for another two centuries. According to the famous vector researcher, medical entomologist Milan Daniel, at the peak of the disease in Constantinople (now Istanbul, Turkey), up to ten thousand people died every day. Based on modern research, scientists have concluded that half of Europeans died long before the plague left the thinned and exhausted population.
The second pandemic, best known as the Black Death, began its attack on Europe in 1346. Introduced from Eastern China according to the Great silk road to Crimea, the infection quickly made its way north, and within four years managed to claim the lives of more than 25 million people, which at that time amounted to a third of the entire European population. The disease did not give up for four centuries - it either hid or flared up (mainly in large cities), wandering from country to country. In 1665-1666 one in five Londoners died from the bubonic plague; but, despite the deplorable situation, medicine remained powerless.
Faces of Plague
Currently, the main forms of plague are bubonic, pulmonary And septic. Occasionally, other varieties are also found: cutaneous, intestinal, pharyngeal and meningeal. Bubonic plague usually develops after the bite of a flea that carries it Y. pestis or after handling carcasses of infected animals and is characterized by inflammation lymph nodes with the formation of their painful conglomerates - “buboes”, “cones” different sizes. If not treated in time, the infection can generalize: sepsis (secondary septic plague) or pneumonia (secondary pneumonic plague) develops. In the latter case, the sputum serves as an infectious substance for other people, who subsequently develop primary pneumonic plague. It is because of the ability to be transmitted from person to person (by airborne droplets) that the pulmonary form of the disease causes the greatest concern. Septic and pulmonary forms sometimes proceed at lightning speed, and death cannot be avoided without urgent antibiotic therapy.
It is possible that the predominance of any form of plague (preferential damage to one or another organ) during various epidemics is associated not only with the mechanisms of transmission of bacteria, but also with the properties of a particular strain-causative agent.
Victim of mutation
It has been known for quite some time that the ancestor of the plague bacillus is Yersinia pseudotuberculosis- enteropathogen, causative agent of pseudotuberculosis. Presumably Y. pestis separated from its ancestral species about twenty thousand years ago. The reason for the evolution was a sharp climate change in the late Pleistocene (the Quaternary period of the Cenozoic era): cold was replaced by heat, causing a restructuring of ecosystems; As a rule, such “jumps” in weather stimulate the evolution of species. However, until recently, the genetic changes critical for the transformation of a pathogen that causes an infectious intestinal disease into a dangerous microorganism that can infect the lungs and provoke fulminant sepsis were not known.
Evolutionary branch Y. pestis was studied by scientists from Northwestern University in the USA. In a study by Wyndham Lathem and Daniel Zimbler, they found that the acquisition of a single gene transformed early forms Y. pestis, already somewhat different genetically and phenotypically from Y. pseudotuberculosis, into a successful pulmonary pathogen. To identify the mechanism of “relocation” of the plague bacillus from the intestines to the lungs, the authors conducted experiments on ancient strains of bacteria and analyzed their behavior in the body of mice. When comparing strains that cause pneumonic plague with relatively harmless ancestors, only one, but very significant, difference was revealed: acquisition of the surface gene became critical squirrel Pla (cm. box) as part of the pPCP1 plasmid. To test the hypothesis, this gene was introduced into the DNA of evolutionarily earlier strains - and the results confirmed the involvement of the Pla protease in effective damage to the respiratory tract.
However, this acquisition Y. pestis it was not enough to learn how to cause the most dangerous systemic infection (septic form of plague). It turned out that such an improvement required only one (!) amino acid substitution in the Pla protein - I259T . This substitution optimized the proteolytic activity of the protein and significantly increased the invasive potential of bacteria during the development of bubonic plague. Thus, scientists believe that first of all, the bacterium acquired the properties of a pulmonary pathogen, a provocateur outbreaks pneumonic plague, and later, as a result of an additional mutation, even more dangerous strains appeared that cause pandemic pneumonic septic and bubonic septic plague.
However, among all the cons Y. pestis scientists also find the advantages of her contact with people. In 2014 in the magazine PLoS ONE An article was published by Sharon de Witte from the University of South Carolina, which stated that people who survived the plague pandemic became the owners of more good health. Scientists have examined the remains of people who lived before, during and after the plague, drawing Special attention on the causes of death and the condition of their bones. The results showed that survivors of the epidemic, as well as their descendants, lived on average to 75 years and had enviable immunity.
A little about Pla
Figure 1. Mechanism of prevention of apoptosis by Pla protease Y. pestis. Left- normal Fas signaling when Pla is inactivated, on right- suppression of apoptosis by the “working” protease. FasL- transmembrane protein localized on the surface of lymphocytes; Fas- FasL receptor; Pla- a protease built into the outer membrane of the bacterial cell. Drawing from.
Why is the Pla protease classified as a virulence factor, i.e. how exactly did she help the plague bacillus, which already boasts a rich arsenal of adaptations for thriving in mammals and transmission by fleas? One of Pla's responsibilities is plasminogen activation: the resulting plasmin destroys fibrin clots, which is important, for example, for the spread of bacteria from buboes throughout the body.
Recently, the development of primary pulmonary infection has been linked to a mechanism associated with inactivation of an apoptotic signaling molecule called Fas ligand(FasL). The role of FasL in the cell is determined by its ability to trigger the process of apoptosis. This protein, which spans the membrane of activated cytotoxic T lymphocytes and airway epithelial cells, has an extracellular domain that binds to the FasR receptor on the surface of other cells (mainly lymphocytes, but also hepatocytes, cancer and some others), which, through activation of caspase proteases, 8 and caspase-3/7 trigger apoptosis. This maintains the homeostasis of immunocytes, prevents autoimmune processes and destroys cells expressing foreign antigens.
Pla protease catalyzes the cleavage of the FasL “working” domain in several places and thereby inactivates this protein - both its membrane and soluble forms. Thus, Pla prevents apoptosis and related inflammatory reactions, necessary for a complete immune response, which contributes to the survival of the pathogen in the host body (Fig. 1).
Experiments carried out on mice showed the following: bacteria with normal Pla protease contributed to a decrease in the amount of FasL, which led to rapid colonization of the lungs, while Yersinia with inactivated Pla multiplied more slowly. The described mechanism of suppressing the immune response, according to scientists, can be used by other pathogens, especially causing infections respiratory tract. And this, in turn, opens up new prospects in the fight against such diseases: you can think, for example, about developing Pla inhibitors or introducing additional FasL molecules.
"Taxi home"
Main carrier Y. pestis from rodents to humans is the flea (Fig. 2), and for the insect this is a forced “passenger delivery”, the price of which is the life of the “carrier”.

Figure 2. A flea clings to a rat's fur. The photograph was taken using an electron microscope and staining was applied. Figure from science.nationalgeographic.com.
Fleas are voracious bloodsuckers. An individual's feeding can last from one minute to several hours; some species manage to fill their stomachs to capacity - so much so that they do not even have time to digest their bloody lunch. Perhaps it was this fact that played a cruel joke on the insects, but it could not have come at a more opportune time. Y. pestis.
The plague bacillus enters the flea's body during its feeding and accumulates in the crop, where it begins to multiply intensively. In this case, the bacteria form a kind of biofilm - a multilayered accumulation of cells immersed in an exopolysaccharide matrix. This phenomenon was even called the “plague block.” Thus, when the flea subsequently feeds, blood does not enter the stomach - the insect feels hungry and more often “goes out hunting.” Infected fleas do not live long (this is understandable - you cannot run far without food), but during this time they manage to infect about 15 animals, including humans.
This happens as follows. Since blood does not pass beyond the biofilm, it accumulates in the esophagus and crop. When a flea bites a victim, there is simply nowhere for a new portion of food to go, and part of the insect’s previous meal along with a portion of bacteria Y. pestis gets into the wound. The baby needs only one hour to “bypass” the unfortunate person’s body and, along with the bloodstream, penetrate into the spleen, liver and lungs. Incubation period(the time from the penetration of the pathogen into the body until the first clinical manifestations) lasts from several hours to 12 days. The transmission diagram of the pathogen* is shown in Figure 3.
* - Unfortunately, a person can compete with a flea in the sophistication of the mechanisms of infection spread. The last major plague epidemic took place in Far East in 1910-1911, but local outbreaks still occur - the plague is not completely defeated, another thing is that antibiotics are now available almost everywhere. But how were outbreaks eliminated before the “antibiotic era”? A remarkable story about the fight against the plague in Soviet Hadrut (1930) was told by the brilliant virologist and doctor Lev Zilber in his memoirs “ Operation "ORE"". This is a truly medical (and even a little espionage - due to the special view of the “party and government” on such events) detective story, both exciting and tragic, making you think about the concept of “doctor”, the peculiarities of the era and selfless work contrary to everything (the savagery of the population, the savagery of the country’s leadership, etc.). The cause of the outbreak was then still able to be established. But... what fleas there are! We highly recommend reading it. - Ed.
"I believe it, I don't believe it"
Around Y. pestis There are many rumors and myths floating around. For example, the bacterium was considered the culprit of the “Plague of Athens” - an epidemic that swept through Ancient Athens in the second year of the Peloponnesian War. The influx of refugees into the Greek city caused overpopulation and overcrowding, which undoubtedly contributed to unsanitary conditions: there was no time to monitor hygiene, since the main forces were aimed at achieving military superiority over the enemies. Under these conditions, an epidemic of “plague” arose, perceived by the Greeks as divine punishment for the family curse of the Alcmaeonids. However, modern research proves the non-involvement Y. pestis to an epidemic in Ancient Greece. Using molecular genetic analysis, it was established that the teeth* found in the burials of victims of the Athenian epidemic do not contain DNA of the plague bacillus, but DNA of bacteria is present Salmonella typhi- pathogen typhoid fever.
* - You can read more about how DNA is extracted from teeth in the article “ » .
Further controversy arises around "assistants" in distribution Y. pestis. The disease is transmitted by fleas, and fleas are transmitted by rodents. It was believed that European rats (Fig. 4), once infected with plague, served as a reservoir of infection for several centuries, but this fact is now disputed by Norwegian scientists. Nils Christian Stenseth from the University of Oslo explains that outbreaks of plague should be associated with weather fluctuations: especially warm and humid spring-summer periods are characterized by rapid development of plants and an abundance of food, the number of rodents in such years increases significantly, which means and the plague spreads faster. A study of ancient records of climate change in Europe and Asia during pandemics led to the conclusion that in Europe the onset of epidemics did correspond to favorable natural conditions, but only... in Asia and with a stable delay of about 15 years. This allowed us to conclude that the plague bacillus was not hidden in European rats for many centuries, but was imported by traders from Asia again and again. Is it true, this hypothesis still requires strict scientific confirmation - Stenset plans to conduct genetic analysis the remains of victims of European plague outbreaks and compare the genomes of pathogens;
Plague stick(Yersinia pestis) - a type of gram-negative spore-forming bacteria, facultative anaerobes. The causative agent of bubonic plague, pneumonia (pneumonic plague) and septicemic plague.
Plague of man
In humans, plague is characterized by a sudden rise in temperature and malaise, which may be accompanied by abdominal pain, nausea and vomiting. There are three main forms of plague, depending on the method of infection: The mortality rate for plague if untreated ranges from 63% to 93%. During treatment modern antibiotics- approximately 16%. Timely treatment with antimicrobial drugs such as aminoglycosides, fluoroquinolones or doxycycline significantly increases the likelihood of a favorable outcome.
The mortality rate for plague if untreated ranges from 63% to 93%. During treatment modern antibiotics- approximately 16%. Timely treatment with antimicrobial drugs such as aminoglycosides, fluoroquinolones or doxycycline significantly increases the likelihood of a favorable outcome. In the XIV-XVII centuries in Europe, from the bubonic plague, according to various estimates, from 50 to 75 million people died. The last outbreak of plague was recorded in Madagascar in the summer of 2015.
Human plague in the USA
According to a release from the Centers for Disease Control and Prevention (CDC), USA dated August 25, 2015, between 1 and 17 human cases of plague were reported annually in the United States from 2001 to 2012 (an average of three cases per year). During the period from April 1, 2015 (to August 25, 2015), 11 cases of human plague were reported in 6 states (causative agent Yersinia pestis). 2 cases have been linked to visits to Yosemite National Park in California. 3 patients (ages 16, 52 and 79 years) died. The reason for the sharp increase in plague incidence in the United States in 2015, as noted in the mentioned release, is not clear.Yersinia pestis in ICD-10
Yersinia pestis is mentioned in the International Classification of Diseases ICD-10 in “Class I. Some infectious and parasitic diseases”, in the block “A20-A28 Some bacterial zoonoses”, where there is a three-character heading “A20 Plague” with an explanation, that it includes an infection caused by Yersinia pestis. This category includes four-character categories:
that it includes an infection caused by Yersinia pestis. This category includes four-character categories: - A20.0 Bubonic plague
- A20.1 Cellulocutaneous plague
- A20.2 Pneumonic plague
- A20.3 Plague meningitis
- A20.7 Septicemic plague
- A20.8 Other forms of plague (with interpretation: Abortive plague. Asymptomatic plague. Minor plague)
- A20.9 Plague, unspecified
Did a small mutation in Yersinia pestis change human history?
In a 2015 paper published in the journal Nature Communications, Dr. D. Zimbler and Dr. W. Lathem from Northwestern University (USA) described the only one they discovered genetic trait Yersinia pestis, which fundamentally changed the course of evolution of the microorganism and the history of mankind. In past Yersinia pestis could not cause pneumonic forms of plague. The acquisition of a single gene for the surface protein Pla led to the transformation Yersinia pestis from a pathogen causing intestinal infectious diseases to a microorganism associated with the development of severe and lethal infections respiratory organs.Role Yersinia pestis in The Black Death is discussed. Some argue that the Black Death spread too quickly to be caused Yersinia pestis. The DNA of this bacterium has been found in the teeth of those who died from the Black Death, while testing of the medieval remains of people who died of other causes did not give positive reaction on Yersinia pestis. This proves that Yersinia pestis is at least a concomitant factor in some (perhaps not all) European plague epidemics. It is possible that selection by the plague may have affected the pathogenicity of the bacterium by weeding out the individuals most susceptible to it.
Available in Russia live vaccine based on a non-virulent plague strain.
Genome
Complete genetic sequences are available for various subspecies of the bacterium: KIM strain (from Medievalis biovar), CO92 strain (from Orientalis biovar obtained from a US clinical isolator), Antiqua strain, Nepal516, Pestoides F. KIM strain chromosomes consist of 4,600,755 base pairs , in strain CO92 - 4,653,728 base pairs. Like relatives Y. pseudotuberculosis And Y. enterocolitica, bacterium Y. pestis contains plasmids pCD1. In addition, it also contains plasmids pPCP1 And pMT1, which are not found in other species of the genus Yersinia. The plasmids listed and the pathogenicity island named HPI, encode proteins that are the cause of the pathogenicity of the bacterium. Among other things, these virulence factors are required for bacterial adhesion and injection of proteins into the host cell, bacterial invasion of the host cell, and capture and binding of iron extracted from red blood cells.
Treatment
Since 1947, the traditional first-stage treatment for Y. pestis were streptomycin, chloramphenicol or tetracycline. There is also evidence of a positive outcome from the use of doxycycline or gentamicin.
It should be noted that isolated strains are resistant to one or two of the agents listed above, and treatment, if possible, should be based on their susceptibility to antibiotics. For some patients, antibiotic treatment alone is not enough, and circulatory, respiratory, or renal support may be required.
Write a review about the article "Plague stick"
Notes
- Collins FM. Pasteurella, Yersinia, and Francisella. In: Barron's Medical Microbiology (Barron S et al , eds.). - 4th ed.. - Univ of Texas Medical Branch, 1996. - ISBN.
- - in an article published in the journal "Knowledge-Power" No. 2 for 2002, reflections are given that criticize the connection of the bacterium with the plague.
- Drancourt M; Aboudharam G; Signolidagger M; Dutourdagger O; Raoult D. (1998). "". PNAS 95 (21): 12637–12640.
- Drancourt M; Raoult D. (2002). "". Microbes Infect. 4 : 105–9.
- . Lenta.ru. Retrieved 23-10=2015.
- Daniel L. Zimbler, Jay A. Schroeder, Justin L. Eddy & Wyndham W. Lathem.. nature.com. Retrieved 13-02=2016.
- . Lenta.ru. Retrieved 23-10=2015.
- Simon Rasmussen.. cell.com. Retrieved 13-02=2016.
- Salyers AA, Whitt DD. Bacterial Pathogenesis: A Molecular Approach. - 2nd ed.. - ASM Press, 2002. - ISBN pp207-12.
- Welkos S et al.. (2002). "". Vaccine 20 : 2206–2214.
- title=Vaccine plague live, dry | url=http://www.epidemiolog.ru/catalog_vac/index.php?SECTION_ID=&ELEMENT_ID=476
- Supotnitsky M. V., Supotnitskaya N. S., 2006,
- Supotnitsky M. V., Supotnitskaya N. S., 2006,
- Deng W et al.. (2002). "". Journal of Bacteriology 184 (16): 4601–4611.
- Parkhill J et al.. (2001). "". Nature 413 : 523–527.
- Wagle PM. (1948). "Recent advances in the treatment of bubonic plague." Indian J Med Sci 2 : 489–94.
- Meyer KF. (1950). "Modern therapy of plague." JAMA 144 : 982–5.
- Kilonzo BS, Makundi RH, Mbise TJ. (1992). "A decade of plagued epidemiology and control in the Western Usambara mountains, north-eastern Tanzania." Acta Tropica 50 : 323–9.
- Mwengee W, Butler T, Mgema S, et al.(2006). "Treatment of plague with gentamicin or doxycycline in a randomized clinical trial in Tanzania." Clin Infect Dis 42 : 614–21.
The content of the article
The name is given in honor of A. Yersin. The genus Yersinia includes several species, of which Y. pestis, Y. enterocolitica, and Y. pseudotuberculosis are pathogenic for humans. They are gram-negative, non-spore-forming rods. They are distinguished by biochemical, antigenic and other characteristics.
